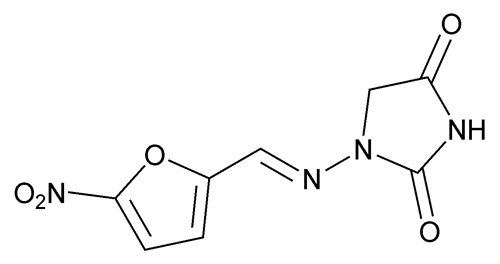
The sustained-release product (macrocrystals-monohydrate) has not been studied, but probably has a delayed peak milk time difficult to predict in individual mothers. Rapid-release capsules (macrocrystals) reach peak milk levels about 1 to 2 hours after the dose with food or 4 hours after a high-fat meal. The time of peak amounts in milk depends on food taken with the drug and dosage form of the drug. The effects in the nursing infant are unknown infants should be observed for possible diarrhea. Although the amount of this drug excreted in breast milk after normal therapeutic doses is considered negligible, the possibility of hemolytic anemia due to immature enzyme systems in the early neonatal period should be considered when using this drug in nursing mothers. According to some authorities: A decision should be made to discontinue breastfeeding or discontinue the drug, taking into account the importance of the drug to the mother.Įxcreted into human milk: Yes (trace amounts) According to some authorities: Use should be temporarily avoided while breastfeeding infants with known/suspected erythrocyte enzyme deficiency (including G6PD deficiency) according to at least 1 manufacturer, caution is recommended while breastfeeding such infants. According to some experts: Use is considered acceptable if infant is healthy and older than 1 month use should be avoided if infant has G6PD deficiency or is younger than 1 month. LactMed: Use is considered acceptable while breastfeeding older infants, but alternative agents are preferred in mothers nursing infants younger than 8 days or infants with glucose-6-phosphate dehydrogenase (G6PD) deficiency at any age. See references Nitrofurantoin Breastfeeding Warnings Pregnancy categories A, B, C, D, and X are being phased out. US FDA pregnancy category Not Assigned: The US FDA has amended the pregnancy labeling rule for prescription drug products to require labeling that includes a summary of risk, a discussion of the data supporting that summary, and relevant information to help health care providers make prescribing decisions and counsel women about the use of drugs during pregnancy. US FDA pregnancy category B: Animal reproduction studies have failed to demonstrate a risk to the fetus and there are no adequate and well-controlled studies in pregnant women. In man, this drug can reduce sperm counts and produce abnormal testicular histology suggestive of arrested spermatogenesis.ĪU TGA pregnancy category A: Drugs which have been taken by a large number of pregnant women and women of childbearing age without any proven increase in the frequency of malformations or other direct or indirect harmful effects on the fetus having been observed. Thus far, studies have not shown a potential for this drug to cause birth defects. There are no controlled data in human pregnancy however, this drug has been in extensive clinical use for many years and its suitability in human pregnancy has been well documented. Risk summary: Malformative risk with use of this drug in pregnant women is unlikely.Īnimal studies have failed to reveal evidence of fetotoxicity or teratogenicity. 
This drug should be used during pregnancy only if clearly needed it should be used at the lowest dose appropriate for specific indication only after careful assessment.ĪU TGA pregnancy category: A (short-term therapy) According to some authorities: Caution is recommended when used at term. According to some authorities: Use is contraindicated in pregnant patients at term (38 to 42 weeks gestation). Use is contraindicated in pregnant patients during labor and delivery, or when onset of labor is imminent. Nitrofurantoin is also known as: Furadantin, Macrobid, Macrodantin, Nitro Macroīecause of the possibility of hemolytic anemia due to immature erythrocyte enzyme systems (glutathione instability): Nitrofurantoin Pregnancy and Breastfeeding Warnings
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
